Olsen group reveals global interplay of ATM and ATR with SUMOylation during replication stress

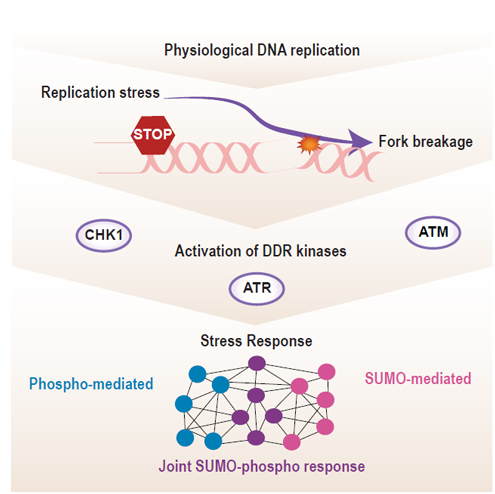
Replication of the human genome is a tremendous task that cells must undertake with great precision to pass on a full and accurate set of genetic information to the next generation of cells. The process of replication is vulnerable to disruptions that are posed by the complex mechanisms that duplicate the genome and by perturbations exerted upon the cells, for example by chemotherapeutics. Cells are equipped with complex networks of proteins that protect the genome against such threats, and an efficient response requires the precise and timely coordination of various post-translational modifications (PTMs) such as phosphorylation of proteins and the enzymes that regulate them. In recent years, protein SUMOylation has emerged as a new important modification in the replication stress response.
In this study published in Cell Reports, the Olsen group together with the group of Alfred Vertegaal from the Leiden University Medical Center investigated the interplay between this SUMOylation and phosphorylation in the replication stress response. Using state-of-the-art mass spectrometry (MS)-based phosphoproteomics and SUMO-proteomics technologies, they identified proteins co-regulated by both modifications. Strikingly, they found that among the most co-regulated proteins were proteins necessary for ATR activation. ATR together with ATM are the central protein kinases that safeguard genomic integrity in response to replication stress and DNA breaks by phosphorylating a plethora of effector proteins. Olsen and colleagues went on to show for the first time, that a global SUMOylation response is instigated when replication is challenged and that this is also regulated by ATR and ATM. This ATM and ATR regulated interplay between phosphorylation and SUMOylation is in particular important at the point of which irreversible damage to the genome occur. This regulation extended to thousands of SUMOylation sites, many of which modifying proteins with known function in protecting the DNA. Combined, the Olsen group has uncovered a new level of integration between phosphorylation and SUMOylation in the cellular systems that protect DNA integrity. This study is of particular interest in the context of cancer therapy due to the increasing efforts to use replication stress induced genomic instability to induce cancer cell killing.
Read the full paper here: Proteomics Reveals Global Regulation of Protein SUMOylation by ATM and ATR Kinases during Replication Stress
Title: Proteomics Reveals Global Regulation of Protein SUMOylation by ATM and ATR Kinases during Replication Stress
Authors: Stephanie Munk, Jo´ n Otti Sigurðsson, Zhenyu Xiao, Tanveer Singh Batth,Giulia Franciosa, Louise von Stechow, Andres Joaquin Lopez-Contreras, Alfred Cornelis Otto Vertegaal, and Jesper Velgaard Olsen (authors from CPR in bold)
