Olsen Group reveals molecular mechanisms explaining functional selectivity of growth factors activating the same receptor
Functional selectivity, also known as biased ligand signaling, is the ligand-dependent selectivity for certain signaling pathways in one and the same receptor. The opposing cellular effects of biased ligands of G-protein coupled receptors are well established, but the idea of functional selectivity has only been scarcely explored in the context of receptor tyrosine kinases (RTKs). The epidermal growth factor receptor (EGFR) is a prominent oncogene that is frequently mutated, amplified and aberrantly activated in cancer and therefore a major drug target in cancer therapy. EGFR can like most other RTKs be activated by binding of different extracellular ligands (growth factors). But in spite of the tremendous amount of work in the field of RTK signaling it is an unsolved biomedical mystery how distinct ligands binding to the same receptor orchestrate different cellular signaling networks to elicit specific outcomes.
The Olsen Group that is part of the Proteomics Program at Novo Nordisk Foundation Center for Protein Research (CPR), together with the Protein Structure and Function Program and the Protein Imaging Platform, and in collaboration with the University of Southern Denmark, deciphered the functional selectivity of growth factors differentially activating the EGF receptor in human cancer cells. The study is published in in this week´s Nature Structural & Molecular Biology.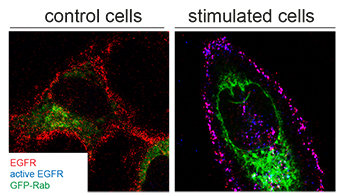
In the study we devised an Integrated Multi-layered Proteomics Approach (IMPA) to systematically examine how biased EGFR ligands affect signaling and cellular outputs. We investigated the cellular responses of the two EGFR ligands, epidermal growth factor (EGF) and transforming growth factor alpha (TGF-alpha), inducing EGFR degradation and recycling, respectively, and uncovered two fine-tuned regulatory molecular mechanisms. By integration of different layers of cellular information (EGFR interactome, ubiquitylome, phosphoproteome and proteome) we refined and prioritized protein candidates regulated in a ligand-dependent manner. In different human cancer cells we demonstrated that EGF receptor degradation depends on EGF-induced phosphorylation of Rab7 on tyrosine 183 and that RCP (also known as RAB11FIP1) recruitment is necessary for EGFR recycling and sustained signaling upon TGF-alpha stimulation. By manipulating the expression level of RCP or phosphorylation of Rab7, EGF receptor trafficking can be rerouted and thereby effectively changing the TGF-alpha specific cellular response to an EGF-like response and vice versa.
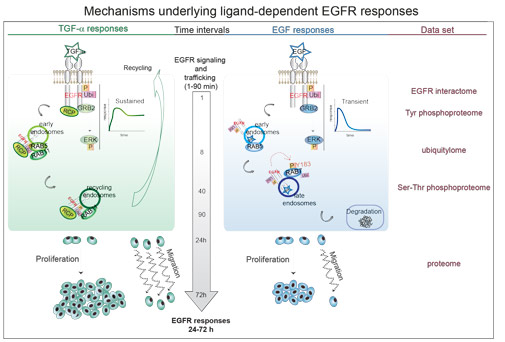
This study not only explains a fascinating conundrum in cell signaling, but it also devises a powerful proteomics approach to pinpoint the key cellular protein regulators of this process. Finally, our research holds potential for re-directing the behaviour of EGFR-expressing cancer cells in human patients, by manipulating ligand-dependent mechanisms underlying cellular responses like proliferation and migration.
For more information, see full text article:
Multilayered proteomics reveals molecular switches dictating ligand-dependent EGFR trafficking
Authors: Chiara Francavilla*, Moreno Papetti, Kristoffer TG Rigbolt, Anna-Kathrine Pedersen, Jon O Sigurdsson, Giuseppe Cazzamali, Gopal Karemore, Blagoy Blagoev, Jesper V Olsen* (Authors from Novo Nordisk Foundation Center for Protein Research in bold).

Novo Nordisk Foundation Center for Protein Research, University of Copenhagen is supported financially by the Novo Nordisk Foundation (Grant agreement NNF14CC0001)
