DNA damage and protein 53BP1's role in DNA repair
New insight into DNA damage
Damage in our DNA can be life-threatening as the DNA carries our genetic information used for growth, development, functioning and reproduction. However, our DNA suffers millions of damaging events each day, coming from internal metabolic processes or external chemical and physical mutagens. The most dangerous form of damage is the so-called DNA double-strand breaks where the entire molecular backbone of the DNA is broken. Fixing such a break is a complex operation and just one wrongly repaired or unrepaired DNA double-strand break can lead to the accumulation of mutations in the body, to cell death, or to serious diseases such as cancer.
In our recent work, we discovered how human cells navigate repair proteins to fix these life-threatening lesions without making mistakes and thereby keep our genome stable.
We identified that a protein called 53BP1 is the critical factor that ensures correct repair of DNA double-strand breaks and show that 53BP1 does this by suppressing a toxic form of repair called single-strand annealing and thereby ensuring that DNA double-strand breaks are repaired via an error-free mechanism called gene conversion.
Protein 53BP1 promotes error free repair of DNA double-strand breaks
We show for the first time that the protein 53BP1 promotes error-free repair of DNA double-strand breaks via gene conversion and that this safeguarding function of 53BP1 plays an important role in breast cancers. Our research has provided a deeper understanding of how healthy human cells fix life-threatening or disease-predisposing genetic lesions without mistakes help us understand how diseases develop. Our research adds to the current efforts within research by converting errors that cancers create while repairing their DNA to specific vulnerabilities – trying to find the cancer’s ‘Achilles heel’ and specifically target it.
For instance, in our recent study we focus on the challenging case of some forms of breast cancers, which do not respond to standard treatments. We show that a subset of tumor cells eliminate the 53BP1 safeguarding mechanism and survive through toxic repair of DNA lesions by single-strand annealing. Understanding the mechanisms of those resistant tumors opens up the possibility to develop drugs against proteins that carry out single-strand annealing, which might resensitize and efficiently treat them.
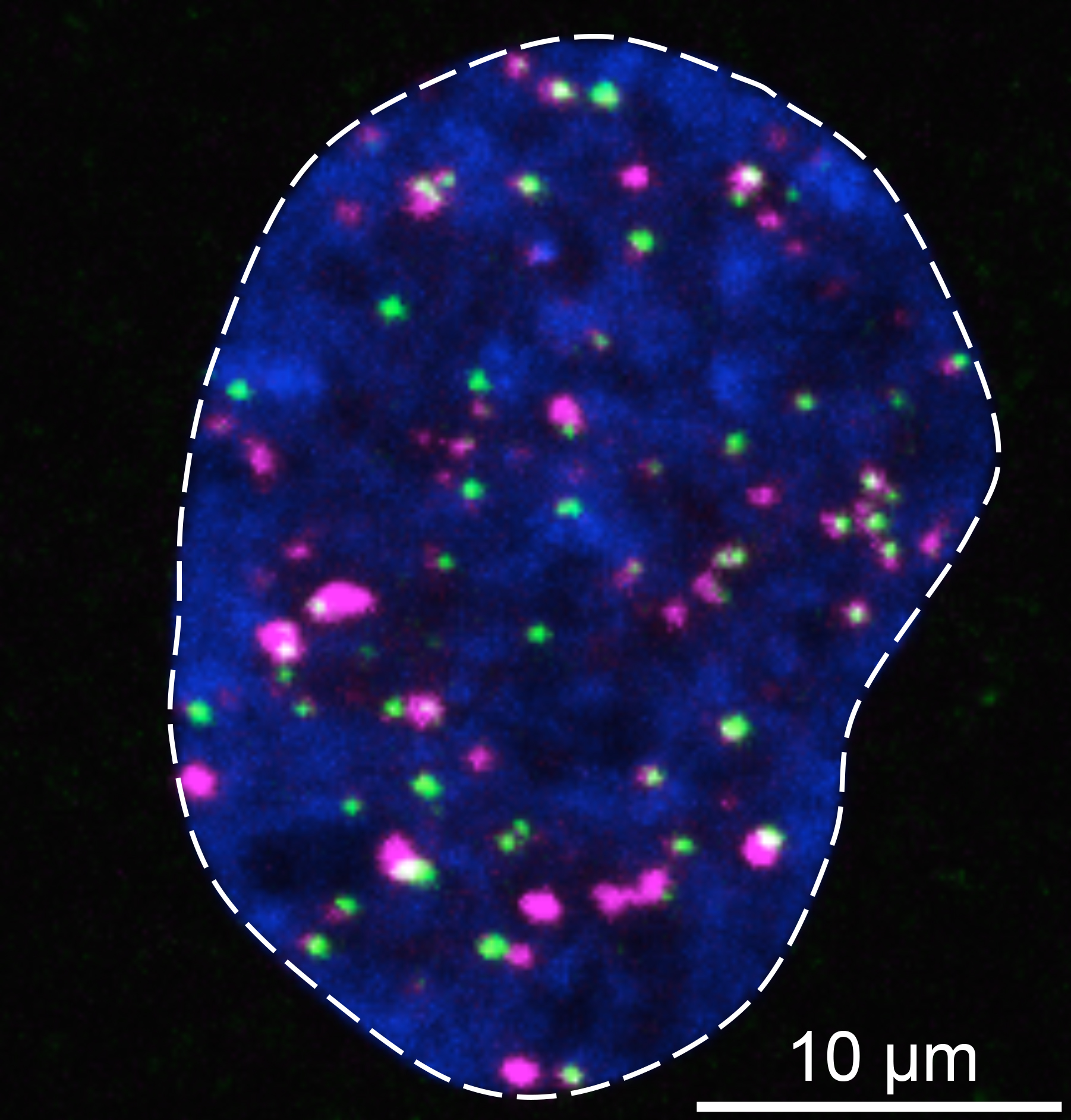
The protein 53BP1 (shown in pink) brings the repair protein RAD51 (shown in green) to DNA double-strand breaks in human cells (nucleus of the cell is shown in blue) to fix those lesions without mistakes.
Background
The research was conducted at Novo Nordisk Foundation Center for Protein Research in Copenhagen in the Lukas group (Chromosome Stability and Dynamics) headed by Professor Jiri Lukas.
The Lukas group is focused on DNA repair mechanisms combining state-of-the-art microscopy with genetics and biochemistry approaches. Our research findings have been generated through close scientific collaboration among the members of our research group. We believe that our success comes from our integrated way of working; combining ideas and experimental techniques to address very exciting (and by nature difficult) questions about the mechanisms that keep our genome free of disease.
Read the paper: 53BP1 fosters fidelity of homology-directed DNA repair
Authors:
Fena Ochs (CPR)
Kuma Somyajit (CPR)
Matthias Altmeyer (Cancer Network Zurich)
Maj-Britt Rask (CPR)
Jiri Lukas (CPR)
Claudia Lukas (CPR)
