Stable MCC binding to the APC/C is required for a functional spindle assembly checkpoint
During cell division the duplicated genetic material in the form of sister chromatids have to be accurately segregated to the two new daughter cells. Failure in this is detrimental and can give rise to cancer and is a major cause of miscarriages. The proper segregation of the sister chromatids is ensured by a surveillance mechanism referred to as the spindle assembly checkpoint (SAC) that delays cells in mitosis when mistakes are sensed. The proteins constituting the SAC have been identified but exactly how this pathway ensures proper chromosome segregation still needs to be defined.
The Nilsson lab now reveals how a key component of the SAC, the mitotic checkpoint complex (MCC), efficiently inhibits mitotic progression through its stable binding to the APC/C complex. The mode of interaction between the MCC and the APC/C is defined using quantitative biochemical approaches. Importantly using time-lapse microscopy and single cell analysis it is shown that specifically interfering with the MCC-APC/C interaction results in a defective SAC. This has important implications for understanding how the SAC ensures proper chromosome segregation and current drug discovery efforts trying to target the APC/C.
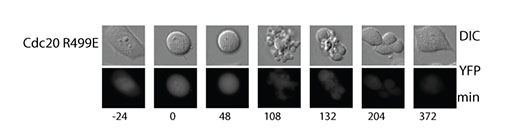
Figure above: Failure in mitotic arrest when cells express Cdc20 R499E hereby preventing stable MCC binding to the APC/C
Article: Stable MCC binding to the APC/C is required for a functional spindle assembly checkpoint
Authors: Jamin B. Hein & Jakob Nilsson
Link to article in EMBO reports
