New insight into cancer defense mechanism published by Nilsson group in Nature Communications
Researchers from Novo Nordisk Foundation Center for Protein Research have identified a new mechanism that ensures that when cells divide, the genomic material passes correctly to the resulting daughter cells. The process, known as chromosome segregation, is vitally important because incorrect passage of the genomic material makes cells prone to develop into cancer cells. The discovery identified by Jakob Nilsson and his research team depends on a protein called BubR1 that is mutated in certain cancers.
Chromosome segregation
The research provides new insight into how our genetic material in form of chromosomes is safely segregated into each of the two new daughter cells that arises when a cell divides. This is a fundamental biological question that has been pursued for more than 100 years because chromosome movements during cell division was some of the earliest structures observed in primitive light microscopes. During cell division, the chromosomes is attached to a remarkable scaffolding structure of proteins, the mitotic spindle, that coordinates the complicated task of ensuring that each new cell in the body receives the correct number of gene copies from its maternal cell.
New function of a known protein
It is known that when cancer cells divide, the genetic material is often not passed on correctly which allows the resulting daughter cells to develop more aggressive growth patterns and become more resistant to chemotherapy. However, the underlying defects are in many cases unknown.
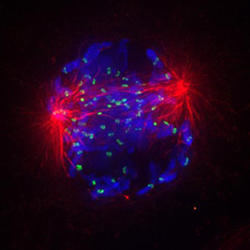
Microscopic view of a human cell undergoing chromosome segregation. Each chromosome (blue) is attached via the mitotic spindle (red) to one of the two poles (red spheres), which will eventually migrate into each of the two new daughter cells together with a complete set of chromosomes.
“There has been a lot of interest in understanding chromosome segregation and also how this is regulated by the protein BubR1. We used sophisticated live cell microscopy to follow chromosome movements in human cells that had mutations in the BubR1 protein, in order to see how this affected chromosome segregation. This allowed us to determine when the cells did not move their chromosomes correctly” says Jakob Nilsson.
“Our work provides a completely new function to how this protein works in human cells, which demonstrates the novel concept that a protein, in this case BubR1, can have more than one function depending on where it is located in the cell” adds Jakob Nilsson.
Potential benefits
The hope is that this research will help in defining better strategies for cancer therapy and provide a better understanding of why chromosomes are often not correctly distributed to the new daughter cells in cancer.
“We know that BubR1 is mutated in certain human cancers so our work could help explain how BubR1 causes cancer when mutated. In the future it could be beneficial to analyze cancer patients for mutations in BubR1. Today, cancer treatment is often based on trial and error but in the future we hope to predict patients’ responses to various drug treatments, based on knowing the mutations present in the individual cancer tissue. Simply put, patients with mutations in BubR1 could be responsive to specific types of drugs and ideally, by analyzing patients for the presence of mutated BubR1, we will know which drugs to use in advance” says Jakob Nilsson, Associate Professor from Novo Nordisk Foundation Center for Protein Research.
For more information, see:
The internal Cdc20 binding site in BubR1 facilitates both spindle assembly checkpoint signalling and silencing
Tiziana Lischetti, Gang Zhang, Garry G. Sedgwick, Victor M. Bolanos-Garcia & Jakob Nilsson
Nature Communications 5, Article number: 5563, DOI: 10.1038/ncomm6563.
Published 08 December 2014.
To read the paper online at Nature Communications, click here.
