Novel insight into cell killing by anticancer agents
Protein phosphatase 2A (PP2A) is a widely expressed enzyme that control a vast number of signalling processes in human cells. PP2A is a known tumor suppressor and given that its function is suppressed in many cancers, there is an interest in developing compounds that can reactivate PP2A for cancer treatment.
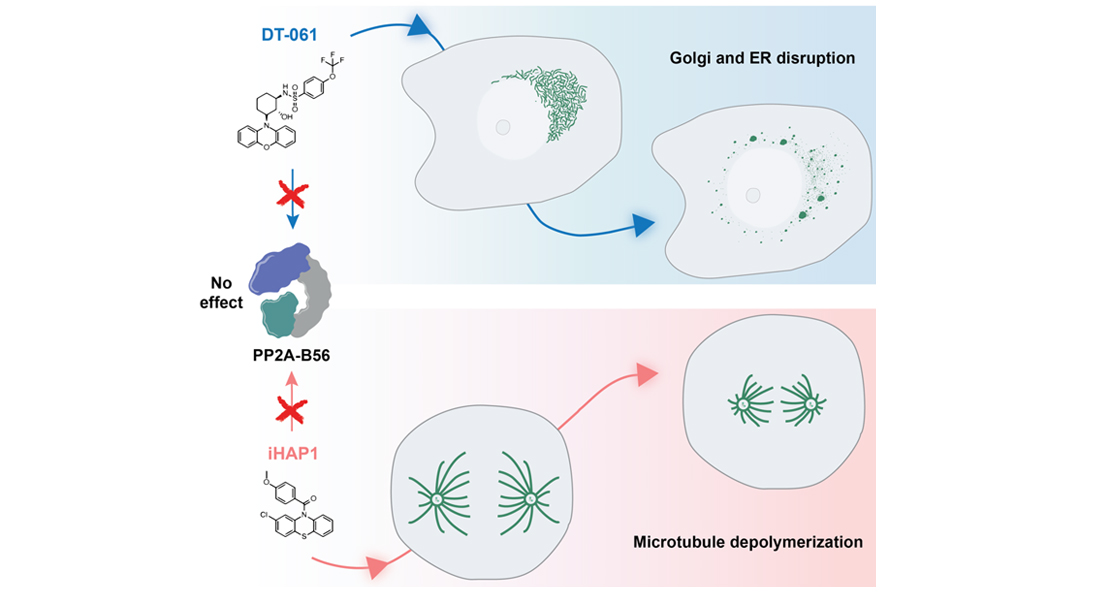
Recent work has suggested that the small molecules iHAP1 and DT-061 can be used to selectively kill cancer cells by reactivating PP2A complexes. However, a new study from the Nilsson group at Novo Nordisk Foundation Center for Protein Research (CPR), University of Copenhagen, provides new insight into the mechanism behind the molecules and argue that this is independent of PP2A activation.
Multi-disciplinary approach revealed the mechanism
When the Nilsson group failed to document an effect of iHAP1 or DT-061 on PP2A, they teamed up with collaborators at the Danish Cancer Society and the Mailand and Montoya groups at CPR, amongst others. First, they undertook a genome wide CRISPR/Cas9 screen to get an unbiased view of the biological pathways affected by the two compounds. Rather than having an effect on PP2A, the screen results indicated that the two drugs affected completely different cellular systems, specifically the Golgi apparatus (for DT-061) and the cytoskeleton (iHAP1).
’We were surprised when we discovered that the ability of iHAP1 and DT-061 to kill cancer cells does not involve PP2A activation. This illustrates how important it is to test the mechanism of action of anticancer reagents. Knowing their molecular targets is essential to understand their mode of action and guarantee the safest and most specific use of anticancer therapies in the clinic’ says Professor Jakob Nilsson from CPR.
The study Chemogenetic profiling reveals PP2A-independent cytotoxicity of proposed PP2A activators iHAP1 and DT-061 was published in The EMBO Journal.
Contact
Professor Jakob Nilsson
+45 35 32 50 53
jakob.nilsson@cpr.ku.dk
